BCH 341 – ASU ONLINE – iCourse – Spring 2018
J.P. Allen, BioPhysical Chemistry, Wiley-Blackwell, 2008. (free PDF version for ASU students)
Chapter 7: problems 1, 8, 13, 16 & 19.
Additional Problems
5-1A: The decomposition of N2O5 is an important process in tropospheric chemistry. The half-life for the first-order decomposition of this compound is 2.1×10^4 sec. How long will it take for a sample of N2O5 to decay to 50% of its initial value?
5-2A: Carbon-14 is a radioactive nucleus with a half-life of 5760 years. Living matter exchanges carbon with its surroundings (for example, through CO2) so that a constant level of carbon-14 is maintained, corresponding to 15.3 decay events per minute. Once living matter has died, carbon contained in the matter is not exchanged with the surroundings, and the amount of carbon-14 that remains in the dead material decreases with time due to radioactive decay. Consider a piece of fossilized cartilage that has 2.4 carbon-14 decay events per minute. How old is this fossilized sample?
5-3: Consider the following sequential reaction scheme:
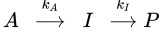
(A) Plot the concentration profiles ([A], [I] and [P] versus time) for the above sequential reaction where the rate constant for A→I is 0.1 inverse seconds and is twice the rate of I→P (i.e., kA = 2kI = 0.1 s^-1).
(B) Determine the time at which [I] is at a maximum for the above sequential reaction, i.e., kA = 2kI = 0.1 s^-1
(C) Assuming that only reactant A is present at t=0, (A) what is the expected time dependence of [P] using the steady-state approximation?
5-4: In acidic conditions, benzyl penicillin (BP) undergoes the following parallel reaction:

In the molecular structures, R1 and R2 indicate alkyl substituents. In a solution where pH=3, the rate constants for the processes at 22C are

(A) What is the yield for P1 formation?
(B) The temperature dependence of the acid-catalyzed hydrolysis of penicillin is investigated, and the dependence of k1 on temperature is given in the following table:

What is the activation energy for this branch of the hydrolysis reaction?
5-5: Proflavine has been used as a bacterial disinfectant and topical antiseptic. However, its use has been limited because it is also a mutagenic compound arising from its ability
![]()
to intercalate between nucleic-acid base pairs in DNA. Although the monomeric form of proflavin can intercalate, the dimer cannot. The monomer and dimer forms of proflavine exist in equilibrium in solution and has been studied by Turner et al. [Nature 239 (1972), 215]:
![]()
who determined that the dimerization rate constant was 8×10^8 1/Ms and the rate constant for dissociation of the dimer back to monomers was 2.0×10^6 1/s.
![]()
In addition, the apparent rate constant for the relaxation of this system toward equilibrium is equal to:
![]()
If 0.10 mM of monomer is present at equilibrium, (A) what is the equilibrium constant for the dimerization reaction, and (B) how long does it take for equilibrium to be achieved?